Photo-induced monomer/dimer kinetics in methylene blue degradation over doped and phase controlled nano-TiO2 films†
Abstract
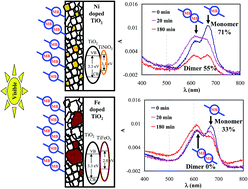
Transparent TiO2 thin films containing 1 to 6 wt% of Ni, Fe and Nb doping were synthesized via a hybrid non-aqua sol–gel dip-coating technique. Metal-doping is found to affect the phase formation, polymorphic transition, visible light absorbance and optical transparency of the TiO2 films. Physico-chemical characterization revealed that, in the given doping range, addition of Ni and Fe enhances the visible light absorbance, while Nb doping does not alter the electronic structure of TiO2. Higher doping concentrations (4 and 6 wt% of Ni and Fe, respectively) lead to the crystallization of a MTiO3 (M: Ni or Fe) phase along with anatase and rutile TiO2. On the other hand, higher Nb doping favors the formation of pure anatase TiO2. 6 wt% of Ni and Fe doping induces the reduction in band gap (3.1 and 2.9 eV), whereas Nb does not alter the TiO2 band gap (3.3 eV). In contrast to the low efficiency of undoped TiO2 films, Ni-doped films yields 71% (for monomer) and 55% (for dimer) photocatalytic degradation efficiency towards methylene blue (MB) under visible light irradiation (λ ∼ 420 nm). This demonstrates the selective and preferential photo-dynamics of the MB monomer during the photocatalytic process. The doped films show a significant Incident photon to current conversion efficiency for oxidation of water under wavelength λ ≥ 400 nm. Such superior improvement in the visible light activity of Ni doped TiO2 is attributed to the presence of nanosized NiTiO3 in the nanostructure of the TiO2 film.


 Please wait while we load your content...
Please wait while we load your content...