Chemoenzymatic modification of silk fibroin with poly(2,6-dimethyl-1,5-phenylene ether) using horseradish peroxidase†
Abstract
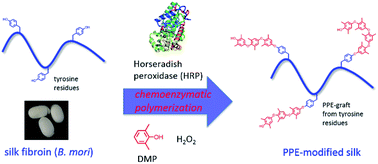
Chemical modification of silk materials is a powerful method for tailoring the desired physical properties for possible application in various fields. In this work, we modified silk fibroin with poly(2,6-dimethyl-1,5-phenylene ether) (PPE) in order to imbue the silks with hydrophobicity so as to resist the absorption of humidity. This modification was achieved by chemoenzymatic polymerization of 2,6-dimethylphenol (DMP) using horseradish peroxidase (HRP) as a catalyst in the presence of silk fibroin obtained from Bombyx mori. The PPE chain content in the modified silk was tuned by varying the feed concentration of DMP. Wide-angle X-ray scattering measurements revealed that β-sheet crystalline structures were formed in the PPE-modified silk, even after the introduction of bulky PPE chains. The PPE-modified silk showed glass transitions derived from the PPE domains, which enabled the formation of self-standing films upon thermal processing. Films of the PPE-modified silk exhibited higher static contact angles of water droplets compared to the native silk films, indicating that the film surface of silk fibroin became more hydrophobic due to the introduction of PPE. These improved physical properties were achieved without sacrificing the inherent secondary structure of silk fibroin, namely, the β-sheet structure that is largely responsible for the mechanical properties of silk materials.

 Please wait while we load your content...
Please wait while we load your content...