Diastereoselective synthesis of aziridines from (1R)-10-(N,N-dialkylsulfamoyl)isobornyl 2H-azirine-3-carboxylates
Abstract
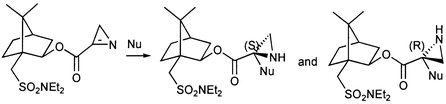
Reaction of the chiral azirine 1a with nucleophiles and dienes is described. Thiols, heteroaromatic nitrogen compounds and phenylmagnesium bromide add to the azirine 1a to give functionalised aziridines 2/3a–h. X-Ray crystal structures for the products 3e and 3g have been obtained. Diastereodifferentiation of the two faces of the azirine 1a is observed in most cases, but only thiophenol gives a single diastereomer (2a). Most mixtures of diastereomers were separated by dry flash chromatography. Benzylamine produced a dimer of the original azirine 1a, compound 9. Representative conjugated dienes (cyclopentadiene, furan and open chain dienes) were added to the chiral azirine 1a. Only poor selectivities were observed. The selectivity was not significantly enhanced in the cycloaddition of cyclopentadiene to the more bulky azirine 1b.

 Please wait while we load your content...
Please wait while we load your content...