Chemoenzymatic formal synthesis of (−)- and (+)-epibatidine†
Abstract
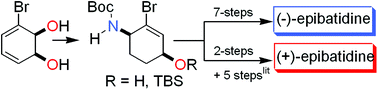
The cis-dihydrocatechol, derived from enzymatic cis-dihydroxylation of bromobenzene using the microorganism Pseudomonas putida UV4, was converted into (−)-epibatidine in eleven steps with complete stereocontrol. In addition, an unprecedented palladium-catalysed disproportionation reaction gave the (+)-enantiomer of an advanced key intermediate employed in a previous synthesis of epibatidine.
 Please wait while we load your content...
Please wait while we load your content...