A surface exposed O-linked galactose residue destabilises the structure of a folded helix–loop–helix dimer†
Abstract
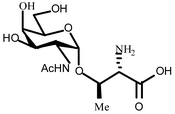
A 42-residue glycopeptide Tn-15 and the corresponding reference polypeptide Thr-15 were designed and synthesized to provide a model system for the study of how glycosylation affects the stability of a molten globule-like protein. Tn-15 and Thr-15 fold into hairpin helix–loop–helix motifs that dimerise to form four-helix bundles and the only difference between the sequences is that Tn-15 carries an O-linked N-acetylgalactosamine residue at the side chain of threonine-15 whereas the sequence Thr-15 is unglycosylated. An analysis of the mean residue ellipticities at 222 nm of the two polypeptides and of the α-H chemical shift deviations from random coil values showed that glycosylation reduced the helical content of the polypeptides and increased the dissociation constant of the helix–loop–helix dimer to form monomers. The pH dependencies of the helical content of Tn-15 and Thr-15 differed as that of Thr-15 was largely unaffected by pH in the range from pH 4 to pH 10, whereas Tn-15 lost almost half of the helical content at pH 4 upon raising the pH to 10. No single amino acid residue was found to ionize in a way that could explain the observed pH dependence of Tn-15. The temperature dependence of the mean residue ellipticity of Tn-15 revealed a surprising decrease in helicity at 278 K in comparison with that at 293 K, reminiscent of cold denaturation, that was not observed for the reference four-helix bundle Thr-15.

 Please wait while we load your content...
Please wait while we load your content...