Novel phenazine fused triphenylene discotic liquid crystals: synthesis, characterisation, thermal, optical and nonlinear optical properties†
Abstract
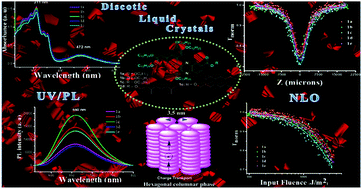
A straightforward synthesis of novel phenazine fused triphenylene discotic liquid crystals (DLCs) is realized. The condensation of 3,4-diaminobenzoic acid with a triphenylene-1,2-diquinone intermediate gives an acid derivative which upon esterification with aliphatic alcohols produces the corresponding monomeric ester derivatives of the phenazine fused triphenylene discotic liquid crystals. The intermediate acid derivative exhibits a rectangular columnar phase along with a high isotropic phase transition temperature. However, the monomeric ester derivatives show a hexagonal columnar mesophase over a broad temperature range and stable when decreased to room temperature upon cooling from the isotropic phase. The mesomorphic properties of the acid and monomeric ester derivatives of the phenazine fused triphenylene DLCs were characterised by polarised optical microscopy (POM), differential scanning calorimetry (DSC) and X-ray diffraction studies (XRD). The molecular structures of the intermediate and final compounds were characterised by spectral and elemental analysis. The photophysical properties of all the monomeric ester compounds were investigated in an anhydrous chloroform solvent and absorption bands around 291–472 nm and the corresponding emission bands at 658–661 nm were observed, respectively. The π-extended conjugation in these materials gives rise to high nonlinear optical properties. These materials have immense potential for use in electro-optical applications.


 Please wait while we load your content...
Please wait while we load your content...