From LCST to UCST: the phase separation behaviour of thermo-responsive polysiloxanes with the solubility parameters of solvents†
Abstract
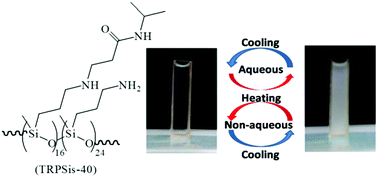
Thermo-responsive polysiloxanes (TRPSis) were synthesized by the catalyst-free aza-Michael addition of poly(aminopropylmethylsiloxane) to N-isopropyl acrylamide. The TRPSis exhibited a lower critical solution temperature (LCST) in water and an upper critical solution temperature (UCST) in several non-aqueous solvents (e.g., acetonitrile, ethylene carbonate, ethyl acetate, butyl acetate, and their mixed solvents). Additionally, the phase separation behaviour of the TRPSis transformed from LCST-type to UCST-type in several water–organic mixed solvents (e.g. water–acetonitrile and water–ethylene carbonate) as the water content decreased. At high water content, the LCST of the TRPSis increased upon the addition of organic solvents (ethanol, acetonitrile, and ethylene carbonate) and the UCST decreased when water was added to the organic solvents, indicating that the LCST and UCST of the thermo-responsive polymers were caused by the synergistic effect of hydrogen bonding and compatibilization. The UCST of the TRPSis shows distinct tendencies using acetonitrile–ethylene carbonate or acetonitrile–ethyl acetate as solvents. In the former case, higher UCST values were observed in the mixed solvent than in their pure one, while lower UCST values were obtained in mixed solvents in the later case. The Hansen solubility parameter was applied to explain the phenomenon and reveal the relationship between the phase separation mechanism of the TRPSis and the solubility parameter of the solvents quantitatively. The smart TRPSis could serve as promising candidates for the all-in-one application of intelligent catalysts for extraction in non-aqueous conditions.


 Please wait while we load your content...
Please wait while we load your content...