Elucidating mechanisms of silica scaling in membrane distillation: effects of membrane surface wettability†
Abstract
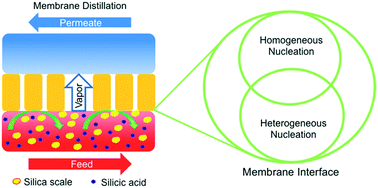
Membrane distillation (MD) has attracted great interest as a promising water purification technology suitable for the treatment of hypersaline wastewater, but its performance is constrained by mineral scaling at the membrane–water interface. Silica scaling is a common but unique type of scaling due to its amorphous nature, but its underlying mechanism has not been elucidated in the literature. In this study, we used a hydrophobic polyvinylidene fluoride (PVDF) membrane, a Janus PVDF membrane with hydrophilic coating, and a superhydrophobic PVDF membrane to investigate the mechanism of silica scaling in MD. The membranes were challenged with silica-containing feedwater with different saturation status. When silica was initially unsaturated, the water vapor flux of MD remained relatively stable, despite the formation of silica scale on the membrane surface. In contrast, a dramatic decline of water vapor flux occurred when the initial silica concentration was supersaturated. However, membrane surface wettability did not affect the starting time of the flux decline. The superhydrophobic membrane exhibited the slowest flux decline rate with time, while the Janus membrane displayed the best water productivity. In colloidal silica fouling that excluded heterogeneous nucleation, the superhydrophobic membrane was superior in both reducing the flux decline and extending water productivity. This difference, along with microscopic characterization of the scaling layer, provided explicit evidence that both homogeneous nucleation and heterogeneous nucleation contribute to the flux decline caused by silica scaling. A four-step mechanism of silica scaling in MD is proposed, in which the decline of water vapor flux is initiated by homogenous nucleation in the bulk solution but facilitated by surface-mediated heterogeneous nucleation.


 Please wait while we load your content...
Please wait while we load your content...