Evaluation of ciprofloxacin destruction between ordered mesoporous and bulk NiMn2O4/CF cathode: efficient mineralization in a heterogeneous electro-Fenton-like process†
Abstract
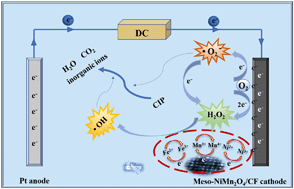
An electro-Fenton-like process was extensively applied to eliminate ciprofloxacin antibiotics pollution in water systems. Here, we developed ordered meso-NiMn2O4 nanoparticles as an efficient electro-Fenton-like catalyst to modify a CF cathode for the degradation and mineralization of ciprofloxacin (CIP). It was found that the carbon felt (CF) cathode modified by the meso-NiMn2O4 catalyst could improve the generation of H2O2, which thereby demonstrated the excellent catalytic property and electrochemical performance. Significantly, the mesoporous architecture of this Fenton-like catalyst provided more activated sites and interconnected channels that could accelerate electron transfer and facilitate the generation of activated radicals (˙OH and ˙O2−). It is worth mentioning that the bifunctional meso-NiMn2O4 catalyst could provide two redox couples (Mn4+/Mn3+ and Ni3+/Ni2+) to enhance the electro-Fenton-like reaction for increasing the generation rate of active radicals. The degradation efficiency of CIP finally reached 100% within 1.5 h by using the meso-NiMn2O4 catalyst, which was better than that of other as-prepared Fenton-like catalysts, such as bulk-NiMn2O4, meso-MnO2, and meso-NiO. Simultaneously, TOC removal of CIP for the meso-NiMn2O4 catalyst was twice than that of bulk-NiMn2O4 catalyst. Finally, a possible heterogeneous electro-Fenton-like reaction mechanism for the efficient destruction of CIP was proposed by using the ordered meso-NiMn2O4 catalyst.


 Please wait while we load your content...
Please wait while we load your content...