Retracted Article: Mutual effect of Cs(i) and Sr(ii) sorption on nano-talc investigated by EXAFS, modeling and theoretical calculations†
Abstract
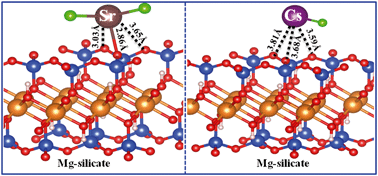
Competitive sorption of Cs(I) and Sr(II) towards nano-talc (a representative magnesium silicate) was investigated by batch experiments, EXAFS, modeling and theoretical calculations. Batch experiments revealed that the decreased extent of Cs(I) sorption in bi-solute was greater than that of Sr(II) sorption, elucidating the preferable sorption of Sr(II) onto nano-talc compared to Cs(I). The high distribution coefficient of Sr(II) was further demonstrated to mean more efficient Sr(II) sorption from groundwater and seawater. The similarity between the adsorbed samples and the aqueous references in the single- and bi-solute indicated outer-sphere complexation of Cs(I) onto nano-talc at low pH by power EXAFS analysis, whereas the formation of Sr inner-sphere mononuclear and bidentate surface complexes was located at the edges of nano-talc platelets at high pH due to the angular dependence of Sr–Si contributions by polarized EXAFS analysis. A diffuse layer model gave satisfactory fits to Cs(I) and Sr(II) sorption onto nano-talc in bi- and single-solute. The high sorption energy and short bond distances, obtained by theoretical calculations, suggested the favorable sorption of Sr(II) onto nano-talc compared to Cs(I). The findings revealed the mutual effect of radionuclides in subsurface environments, which is of great importance for an in-depth understanding of the transportation and fate of radionuclides at the water–mineral interface and for evaluating the potential risk of radionuclides in geological disposal.


 Please wait while we load your content...
Please wait while we load your content...