Temperature-programmed reduction with NO as a characterization of active Cu in Cu-CHA catalysts for NH3-SCR
Abstract
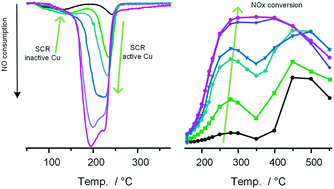
Temperature programmed reduction with NO (NO-TPR) is introduced as a characterization method, by monitoring the reduction of CuII to CuI in a mixture of NH3 and NO. Since the NO-TPR method is based on the oxidation and reduction half cycles of the NH3-SCR reaction, quantitative information about the amount of active Cu in Cu-CHA is obtained. Furthermore, information on the stability and reactivity of reaction intermediates in the NH3-SCR cycle is obtained as well. The reduction of the Cu is followed by monitoring the consumption of NO, after oxidation of the catalyst in O2 or a mixture of NO and O2 to form the CuII state. Two distinct states of the Cu are revealed. The first state, corresponding to the reduction of a Cu-oxide species, is reduced around 130 °C and is observed at low Cu content only. The second state corresponds to the reduction of a Cu-nitrate species around 200–230 °C. The low-temperature activity of Cu-chabazite catalysts with low Cu content show the same trend as the Cu-nitrate species observed in NO-TPR. The fraction of Cu-oxide in NO-TPR decreases with increasing Cu content, leading to a non-linear dependence of the NH3-SCR activity on the Cu-content. At high Cu content, all Cu forms a stable Cu-nitrate species, and the NH3-SCR activity becomes proportional to the Cu content. This agrees well with the known behavior of Cu-CHA catalysts, indicating that NO-TPR seems to be a viable method for the characterization of Cu-CHA materials as NH3-SCR catalysts.


 Please wait while we load your content...
Please wait while we load your content...