The effect of morphology of α-MnO2 on catalytic decomposition of gaseous ozone
Abstract
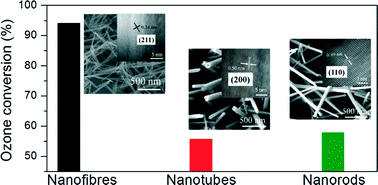
To learn the effect of morphology of α-MnO2 on its catalytic activity for ozone decomposition, three α-MnO2 nanostructures with different morphologies including nanofibres, nanorods and nanotubes were prepared via a hydrothermal process and characterized by XRD, BET, TEM, H2-TPR, O2-TPD, TGA and XPS. The three α-MnO2 show differences in surface area, average oxidation state of Mn and adsorbed surface oxygen species, as well as exposed crystal facets, i.e. (211), (110) and (200) corresponding to nanofibres, nanorods and nanotubes, respectively. It is found that the catalytic activity of α-MnO2 is dependent on the amount of oxygen vacancies. The α-MnO2 nanofibers with the most abundant oxygen vacancies exhibit the best activity, which is ascribed to their largest specific surface area and exposed (211) facet. In situ Raman analysis indicated that intermediate peroxide species formed on the α-MnO2 surface during ozone decomposition, and peroxide species over the α-MnO2 nanofibres are produced at lower Raman band due to their uniform dispersion, which is favorable to ozone decomposition. Finally, the ozone decomposition steps involving oxygen vacancy is proposed.

 Please wait while we load your content...
Please wait while we load your content...