A two-step synthesis of Fe-substituted hexaaluminates with enhanced surface area and activity in methane catalytic combustion
Abstract
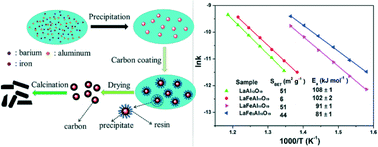
Hexaaluminates have been regarded as one of the most promising candidates for methane catalytic combustion due to their superior thermal stability up to 1600 °C. However, the main challenge for their further application lies in the difficulty in enhancing the surface area. Herein, Fe-substituted hexaaluminates (LaFexAl12−xO19, x = 0–2) with high surface areas (44–51 m2 g−1) have been successfully synthesized via a new two-step method for the first time, in which N,N-dimethylformamide (DMF) was used as a difunctional solvent to derive the precipitant dimethylamine and to control the precipitation rates of the three metal cations, leading to superior homogeneity of the precipitate and thus lowering of the phase formation temperature (lower than 1100 °C), and the resin/carbon coating process was employed to separate the crystallization and grain-growth process, avoiding the aggregation of particles. Methane catalytic combustion tests revealed that both the high surface area and the introduction of Fe could result in low apparent activation energy. The LaFeAl11O19 catalyst exhibited even higher activity than the conventional Pd/Al2O3 catalyst and those prepared by other methods such as the reverse microemulsion method. Mössbauer spectroscopy characterization verified that the catalytic activity of Fe cations should be related to the Fe3+ ions in Al(3) sites and Al(5) sites in the mirror planes.

 Please wait while we load your content...
Please wait while we load your content...