Synergistic effect of Mo2N and Pt for promoted selective hydrogenation of cinnamaldehyde over Pt–Mo2N/SBA-15†
Abstract
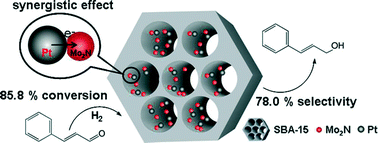
The introduction of a suitable co-catalyst is a promising way to improve the catalytic efficiency of Pt catalysts. Herein, SBA-15 modified with well-dispersed Mo2N nanoparticles (NPs) is synthesized by anchoring phosphomolybdic acid (HPM) on aminated SBA-15 (SBA-15-NH2) primarily, followed by nitridation treatment under NH3. After loading of Pt, the Pt–Mo2N/SBA-15 catalyst with close-contact Pt and Mo2N NPs can be formed. The intensive interaction of Pt and Mo2N benefited from the close contact is verified by XRD and XPS tests. As a result, the ternary Pt–Mo2N/SBA-15 catalysts have shown superior performance to Pt/SBA-15 catalysts for the selective hydrogenation of cinnamaldehyde to cinnamyl alcohol. The TOF over the 1 wt% Pt–Mo2N/SBA-15 catalyst is close to that over the 3 wt% Pt/SBA-15 catalyst, but its selectivity to cinnamyl alcohol (COL) is 2.5 times that of 3 wt% Pt/SBA-15. The promoted activity of Pt–Mo2N/SBA-15 is mainly attributed to the synergistic effect of Pt and Mo2N NPs, which can cause the full development of the catalytic ability of Pt, thus reducing the Pt usage, through increasing the dispersion of Pt NPs and the fraction of Pt0 species in the catalyst.

 Please wait while we load your content...
Please wait while we load your content...