Probing the interaction between solid benzene and water using vacuum ultraviolet and infrared spectroscopy
Abstract
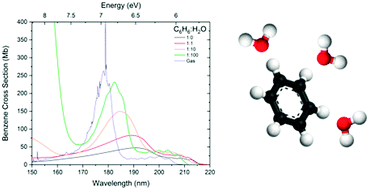
We present results of a combined vacuum ultraviolet (VUV) and infrared (IR) photoabsorption study of amorphous benzene : water mixtures and layers to investigate the benzene–water interaction in the solid phase. VUV spectra of 1 : 1, 1 : 10 and 1 : 100 benzene : water mixtures at 24 K reveal a concentration dependent shift in the energies of the 1B2u, 1B1u and 1E1u electronic states of benzene. All the electronic bands blueshift from pure amorphous benzene towards gas phase energies with increasing water concentration. IR results reveal a strong dOH-π benzene–water interaction via the dangling OH stretch of water with the delocalised π system of the benzene molecule. Although this interaction influences the electronic states of benzene with the benzene–water interaction causing a redshift in the electronic states from that of the free benzene molecule, the benzene–benzene interaction has a more significant effect on the electronic states of benzene. VUV spectra of benzene and water layers show evidence of non-wetting between benzene and water, characterised by Rayleigh scattering tails at wavelengths greater than 220 nm. Our results also show evidence of benzene–water interaction at the benzene–water interface affecting both the benzene and the water electronic states. Annealing the mixtures and layers of benzene and water show that benzene remains trapped in/under water ice until water desorption near 160 K. These first systematic studies of binary amorphous mixtures in the VUV, supported with complementary IR studies, provide a deeper insight into the influence of intermolecular interactions on intramolecular electronic states with significant implications for our understanding of photochemical processes in more realistic astrochemical environments.


 Please wait while we load your content...
Please wait while we load your content...