Dielectric spectra of ionic liquids and their conversion to solvation dynamics: a detailed computational analysis of polarizable systems†
Abstract
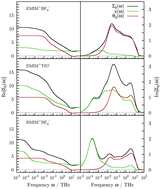
For the three molecular ionic liquids 1-ethyl-3-methylimidazolium tetrafluoroborate, 1-ethyl-3-methylimidazolium trifluoromethanesulfonate and 1-butyl-3-methylimidazolium tetrafluoroborate, dielectric spectra were calculated from molecular dynamics simulations based on polarizable force fields. Using the reaction field continuum model the dielectric spectra were converted to the solvation dynamics of coumarin 153. It is shown in detail that the inclusion of the static conductivity in this model is essential. When simplifying the dielectric spectrum to the static conductivity hyperbola, the solvation response function becomes mono-exponential. Taking into account the frequency dependence of the conductivity, the typical two time-regimes of the solvation response function in ionic liquids are already obtained. However, the mean relaxation time remains the same. When converting the complete dielectric spectrum, i.e. also including frequency-dependent dielectric permittivity, quantitative changes are observed, but the qualitative shape is conserved. In accordance with previous experimental studies, solvation dynamics in ionic liquids predicted by the reaction field continuum model is too fast for longer times. This correlates with the suppression of the fine structure of the dielectric spectrum at low frequencies by the static conductivity hyperbola. By scaling down the static conductivity this effect can be partially amended. In addition to the impact of the solvent dielectric spectrum on solvation dynamics, solute-specific effects, i.e. anisotropy in shape and charge distribution as well as polarizability, were also studied.

 Please wait while we load your content...
Please wait while we load your content...