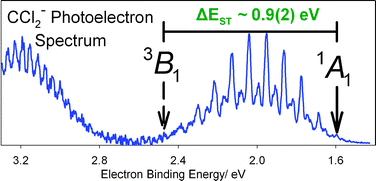
We report new 351 nm negative ion photoelectron spectra of CCl2−, CBr2−, and CI2−. This study was undertaken in an attempt to understand the major discrepancy between dihalocarbene (CX2, X = Cl, Br, I) singlet–triplet splittings reported by our laboratory (R. L. Schwartz, G. E. Davico, T. M. Ramond, W. C. Lineberger, J. Phys. Chem. A., 1999, 103, 8213) and new theoretical values. Our recent experiments show that a dihalomethyl anion (CHX2−) contaminant in the dihalocarbene anion beam, previously considered insignificant, made a major contribution to the reported CX2− photoelectron spectra. Thus, the interpretations of the earlier CX2− spectra and the reported singlet–triplet splittings were incorrect. Replacing O− with OH− in the anion formation process yields a pure dihalomethyl anion, whose highly structured photoelectron spectrum can be subtracted from the contaminated spectrum, yielding a clean CX2− photoelectron spectrum. The new CCl2− photoelectron spectrum displays resolved vibronic transitions to the two lowest electronic states of CCl2: X1A1 and a3B1. The electron affinity of X1A1 CCl2 is 1.593(6) eV. A large change in geometry between the anion and the neutral triplet state precludes the direct observation of the triplet origin. The energy difference between the X1A1 and a3B1 states of CCl2 is estimated to be ∼0.9(2) eV, consistent with high-level theoretical studies. While we confirm similar dihalomethyl anion contaminants in the earlier photoelectron spectra of CBr2− and CI2− and report new photoelectron spectra for these ions, the paucity of resolved features in the spectra provides limited additional thermochemical information.

 Please wait while we load your content...
Please wait while we load your content...