Quantitative analysis of iontophoretic drug delivery from micropipettes
Abstract
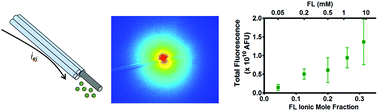
Microiontophoresis is a drug delivery method in which an electric current is used to eject molecular species from a micropipette. It has been primarily utilized for neurochemical investigations, but is limited due to difficulty controlling and determining the ejected quantity. Consequently the concentration of an ejected species and the extent of the affected region are relegated to various methods of approximation. To address this, we investigated the principles underlying ejection rates and examined the concentration distribution in microiontophoresis using a combination of electrochemical, chromatographic, and fluorescence-based approaches. This involved a principal focus on how the iontophoretic barrel solution affects ejection characteristics. The ion ejection rate displayed a direct correspondence to the ionic mole fraction, regardless of the ejection current polarity. In contrast, neutral molecules are ejected by electroosmotic flow (EOF) at a rate proportional to the barrel solution concentration. Furthermore, the presence of EOF was observed from barrels containing high ionic strength solutions. In practice, use of a retaining current draws extracellular ions into the barrel and will alter the barrel solution composition. Even in the absence of a retaining current, diffusional exchange at the barrel tip will occur. Thus behavior of successive ejections may slightly differ. To account for this, electrochemical or fluorescence markers can be incorporated into the barrel solution in order to compare ejection quantities. These may also be used to provide an estimate of the ejected amount and distribution provided accurate use of calibration procedures.


 Please wait while we load your content...
Please wait while we load your content...