Ferrite-based materials for anticorrosion: comparative study of ZnFe2O4, CuFe2O4, and SrFe12O19
Abstract
Three chitosan ferrite nanoparticles-ZnFe2O4, CuFe2O4, and SrFe12O19-were synthesized via an ultrasound-assisted PEG route, yielding phase-pure products with controlled morphologies. Structural and compositional analyses confirmed phase purity, tailored morphology, and mesoporosity across systems for cubic spinel ZnFe2O4 and cuprospinel CuFe2O4. In contrast, SrFe12O19 exhibited coexistence of hexagonal M-type ferrite and rhombohedral α-Fe2O3, indicating a multiphase nature. These nanoparticles were incorporated into chitosan-ferrite nanocomposites and evaluated as corrosion inhibitors for carbon steel in 1.0 M HCl via potentiodynamic polarization (PDP), electrochemical frequency modulation (EFM), and electrochemical impedance spectroscopy (EIS). All of the techniques revealed concentration-dependent inhibition, with ZnFe2O4-based composites consistently demonstrating the highest efficiencies (up to 98.73% by PDP, 94.35% by EFM, and 97.38% by EIS). The inhibition mechanism was identified as mixed-type, primarily affecting the cathodic reaction, supported by causality factors and impedance parameters such as increased Rct and decreased Cdl. Adsorption modeling showed strong monolayer behavior with spontaneous composite–metal surface interaction, validated by Langmuir fitting and 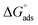 values (∼−28 kJ mol−1). These results highlight the performance of ultrasound-engineered ferrite nanocomposites in forming protective films and reducing acid-induced corrosion, underscoring their application potential in advanced coating systems.
values (∼−28 kJ mol−1). These results highlight the performance of ultrasound-engineered ferrite nanocomposites in forming protective films and reducing acid-induced corrosion, underscoring their application potential in advanced coating systems.



 Please wait while we load your content...
Please wait while we load your content...