Photosensitizer-free, visible light-mediated recyclable gold-catalyzed cross-coupling of aryldiazonium salts and alkynyltrimethylsilanes†
Abstract
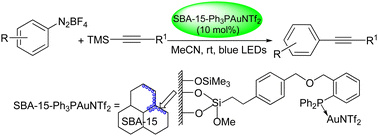
A new mesoporous SBA-15-anchored triphenylphosphine–gold(I) complex [SBA-15-Ph3PauNTf2] was synthesized via the condensation of (4-(chloromethyl)phenethyl)trimethoxysilane with SBA-15, followed by reaction with (2-(diphenylphosphino)phenyl)methanol and Me2SAuCl and then treatment with AgNTf2 and characterized by different physico-chemical techniques. In the presence of 10 mol% of SBA-15-Ph3PAuNTf2, the photosensitizer-free cross-coupling reaction of aryldiazonium tetrafluoroborates and alkynyltrimethylsilanes proceeded smoothly in acetonitrile under irradiation with blue LEDs at room temperature to afford diverse arylalkynes in moderate to good yields with high functional group tolerance, including aryl halides incompatible with traditional cross-coupling. This new heterogenized gold(I) catalyst could be easily recovered through a simple centrifugation process and reused at least nine times without any significant drop in its catalytic efficiency.


 Please wait while we load your content...
Please wait while we load your content...