Validity of the Onsager–Glarum relationship in a molecular coulomb fluid: investigation via temperature-dependent molecular dynamics simulations of a representative ionic liquid, [BMIM][PF6]†
Abstract
Temperature dependent (T = 298–450 K) molecular dynamics simulations were carried out with the ionic liquid, 1-butyl-3-methylimidazolium hexafluorophosphate ([BMIM][PF6]), in order to explore the validity of the Onsager–Glarum (OG) relationship, 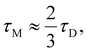 for dipolar liquids with τM andτD being respectively the molecular rotation and the dielectric relaxation times. The presence of strong molecular correlations and the inherent spatiotemporal heterogeneity in [BMIM][PF6] is expected to severely limit the validity of the OG relationship which is based on the continuum model point dipole description of dipolar liquids. We explored this aspect via simulations of the rank (
for dipolar liquids with τM andτD being respectively the molecular rotation and the dielectric relaxation times. The presence of strong molecular correlations and the inherent spatiotemporal heterogeneity in [BMIM][PF6] is expected to severely limit the validity of the OG relationship which is based on the continuum model point dipole description of dipolar liquids. We explored this aspect via simulations of the rank ( ) dependent collective single particle reorientational correlation functions (C
) dependent collective single particle reorientational correlation functions (C (t)), the structural hydrogen bond fluctuation dynamics (CHB(t)), and the medium viscosity (η). The simulated average single particle reorientational correlation time (τ
(t)), the structural hydrogen bond fluctuation dynamics (CHB(t)), and the medium viscosity (η). The simulated average single particle reorientational correlation time (τ ) was used as a bridge between τM and τD in order to explore the validity of the OG relationship in [BMIM][PF6]. The coupling between the simulated 〈τ
) was used as a bridge between τM and τD in order to explore the validity of the OG relationship in [BMIM][PF6]. The coupling between the simulated 〈τ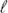 〉 and simulated η was then followed to reflect the medium heterogeneity. Simulated CHB(t) showed strongly stretched exponential relaxations with β (stretching exponent) ∼0.2–0.5. The ratio between 〈τ
〉 and simulated η was then followed to reflect the medium heterogeneity. Simulated CHB(t) showed strongly stretched exponential relaxations with β (stretching exponent) ∼0.2–0.5. The ratio between 〈τ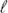 =1〉 and 〈τ
=1〉 and 〈τ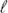 =2〉 showed significant deviation from the Debye
=2〉 showed significant deviation from the Debye 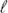 (
(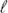 + 1) law. The simulated Kirkwood g factor, being significantly less than unity, indicated strong orientational correlations among the molecular ions. Estimated hydrodynamic effective volumes, (Veff), from 〈τ
+ 1) law. The simulated Kirkwood g factor, being significantly less than unity, indicated strong orientational correlations among the molecular ions. Estimated hydrodynamic effective volumes, (Veff), from 〈τ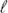 〉 were found to be considerably smaller than the molecular volume of the cation, indicating either small amplitude rotation of the dipolar ion or a non-Brownian move such as angular jumps.
〉 were found to be considerably smaller than the molecular volume of the cation, indicating either small amplitude rotation of the dipolar ion or a non-Brownian move such as angular jumps.
![Graphical abstract: Validity of the Onsager–Glarum relationship in a molecular coulomb fluid: investigation via temperature-dependent molecular dynamics simulations of a representative ionic liquid, [BMIM][PF6]](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3NJ01996A&imageInfo.ImageIdentifier.Year=2023)


 Please wait while we load your content...
Please wait while we load your content...