Syngas production at a near-unity H2/CO ratio from photo-thermo-chemical dry reforming of methane on a Pt decorated Al2O3–CeO2 catalyst†
Abstract
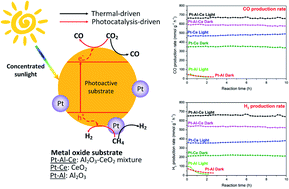
In this work, a Pt catalyst supported on an equimolar Al2O3–CeO2 binary oxide (Pt–Al–Ce) was prepared and applied in photo-thermo-chemical dry reforming of methane (DRM) driven by concentrated solar irradiation. It was found that the Pt–Al–Ce catalyst showed good stability in DRM reactions and significant enhancements in H2 and CO production rates compared with Pt/CeO2 (Pt–Ce) and Pt/Al2O3 (Pt–Al) catalysts. At a reaction temperature of 700 °C under 30-sun equivalent solar irradiation, the Pt–Al–Ce catalyst exhibits a stable DRM catalytic performance at a H2 production rate of 657 mmol g−1 h−1 and a CO production rate of 666 mmol g−1 h−1, with the H2/CO ratio almost equal to unity. These production rates and the H2/CO ratio were significantly higher than those obtained in the dark at the same temperature. The light irradiation was found to induce photocatalytic activities on Pt–Al–Ce and reduce the reaction activation energy. In situ diffuse reflectance infrared Fourier transform spectroscopy (in situ DRIFTS) was applied to identify the active intermediates in the photo-thermo-chemical DRM process, which were bidentate/monodentate carbonate, absorbed CO on Pt, and formate. The benefits of the binary Al2O3–CeO2 substrate could be ascribed to Al2O3 promoting methane dissociation while CeO2 stabilized and eliminated possible coke formation, leading to high catalytic DRM activity and stability.


 Please wait while we load your content...
Please wait while we load your content...