Practical chemoselective aromatic substitution: the synthesis of N-(4-halo-2-nitrophenyl)benzenesulfonamide through the efficient nitration and halogenation of N-phenylbenzenesulfonamide†
Abstract
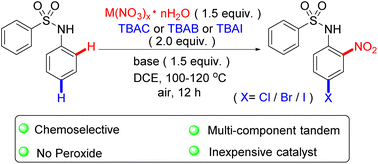
A novel route involving the metal-promoted tandem nitration and halogenation of N-phenylbenzenesulfonamide to synthesize N-(4-halo-2-nitrophenyl)benzenesulfonamide derivatives has been developed. The method shows highly practical chemoselective and functional group compatibility. In addition, it employs insensitive and inexpensive Cu(NO3)2·3H2O, Fe(NO3)3·9H2O, and NH4NO3 as the nitration reagents, and it provides a direct approach for the preparation of 4-halo-2-nitroaniline, which is a crucial intermediate for the synthesis of benzimidazoles and quinoxaline derivatives.


 Please wait while we load your content...
Please wait while we load your content...