Fabrication of copper supported porous silica–alumina hollow spheres for catalytic decomposition of nitrous oxide
Abstract
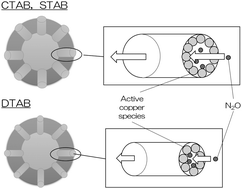
Copper supported porous silica–alumina hollow sphere catalysts were prepared using surfactant micelles to control the size distribution of interparticle spaces in the hollow sphere shells. The specific surface areas and pore volumes of the unsupported and copper supported hollow spheres tended to increase with increasing the ratio of deionized water to ethyl alcohol, and the active copper ion species was well dispersed on the hollow spheres as observed from the results of UV-vis spectroscopy. The copper supported hollow sphere catalyst prepared with a high ratio of deionized water to ethanol tended to show high activity for decomposition of nitrous oxide at reaction temperatures from 473 to 773 K. The present study also investigated the influence of the carbon number of surfactant molecules on the size of the interparticle spaces in the hollow sphere shells and the catalytic activity of the copper supported silica–alumina hollow spheres for decomposition of nitrous oxide. The size of interparticle spaces in the hollow sphere shells decreased with decreasing the carbon number, and the catalytic activity of the copper supported hollow sphere catalyst prepared with DTAB (carbon number = 12) showed the highest activity even though dispersion of the active copper ion species was not so high. These results indicate that the catalytic activity of the copper supported silica–alumina hollow sphere catalyst was influenced not only by the dispersion of the active copper species but also by the size of the interparticle spaces in the hollow sphere shells depending on the contact time of the nitrous oxide reactant with the active copper species.


 Please wait while we load your content...
Please wait while we load your content...