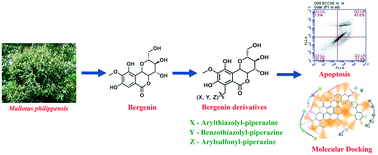
Piperazine tethered bergenin heterocyclic hybrids: design, synthesis, anticancer activity, and molecular docking studies†
Abstract
In an attempt to develop natural product-based anticancer agents, a series of novel piperazine-linked bergenin heterocyclic hybrids bearing arylthiazolyl (5a–e), benzothiazolyl (10a–i), and arylsulfonyl (13a–o) were synthesized using the classical Mannich reaction and evaluated for their anticancer activity. All the synthesized derivatives were assessed for in vitro cytotoxic activity against a panel of human cancer and normal cell lines and the results showed that most of the compounds exhibited significant cytotoxic activity against cancer cells and mild cytotoxicity against normal cells. In particular, the compounds 5a, 5c, 10f, and 13o showed potent cytotoxic activity against tongue and oral cancer cell lines compared to the parent compound (<100 μM). Considering the efficacy, the compounds 5a, 5c, 10f, and 13o were subjected to cell cycle analysis and the results indicated that the compounds mitigated the cell cycle progression at the G0/G1 phase in the tongue and oral cancer cell lines. Subsequently, the annexin V/PI staining assay demonstrated that the compounds 5a, 5c, 10f, and 13o induced early and late apoptosis against tongue cancer and necrosis against oral cancer. Further, gene expression analysis revealed that 5a, 5c, and 13o treatment regulated the BAX and BcL-2 expression and also the selected compounds significantly reduced the expression level of vimentin, oct-4, and nanog. In addition, molecular docking studies revealed that the selected derivatives have strong binding energy with the BcL2 protein and downregulates the expression. Taken together, the study results implied that these compounds are promising anticancer candidates by modulating the epithelial to mesenchymal transition axis and could be considered for further development of novel anticancer drugs.

 Please wait while we load your content...
Please wait while we load your content...