Rhodium catalysts with superbulky NHC ligands for the selective α-hydrothiolation of alkynes†
Abstract
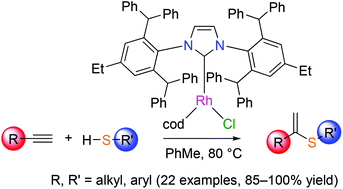
Eight rhodium complexes—including four new compounds—with the generic formula [RhCl(cod)(NHC)] (cod is 1,3-cyclooctadiene) differing by the size of their N-heterocyclic carbene (NHC) ligand were prepared, characterized, and found to be catalytically active in the hydrothiolation of terminal alkynes with aliphatic or aromatic thiols. The steric bulk of the carbene was found to markedly influence the reaction rate and selectivity. In particular, superbulky NHCs led to the almost quantitative formation of the sole α-vinyl sulfide products. The experimental conditions were optimized to allow the straightforward synthesis of a broad range of mono- and disubstituted α-adducts starting from terminal alkynes (18 examples) and thiols (5 examples). Altogether, the procedure devised in this study provides an easy access to α-vinyl sulfides with full atom economy and a low catalyst loading.


 Please wait while we load your content...
Please wait while we load your content...