Stepwise on-surface synthesis of thiophene-based polymeric ribbons by coupling reactions and the carbon–fluorine bond cleavage†
Abstract
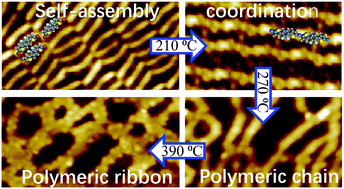
The rational synthesis of thiophene-based cross-coupled polymers on surfaces has been attracting more attention recently. Here, we report the stepwise activation of 5,5′-(2,3-difluoro-1,4-phenylene)bis(2-bromothiophene) as a precursor to synthesize thiophene-based polymeric ribbons on the Au(111) surface. Scanning tunneling microscopy studies showed that the precursor adopted different conformations in the self-assembled structure, organometallic species, and covalent polymers. On annealing the sample at a relatively low temperature (150 °C), the conversion of the organometallic structure into a covalent product with straight lines was observed, in which the Br adatoms arranged between the neighboring chains. On further annealing the sample at 270 °C, the detached Br adatoms played a key role in promoting the C–H bond activation. The cross-linked polymer was achieved by a combination of Ullmann and dehydrogenative coupling. When the annealing temperature was up to 390 °C, the C–F bond activation was triggered, which led to the formation of polymeric ribbons resulting from the cyclodehydrogenation of the fluorinated polymer. This study further supplements the reaction mechanism of thiophene-based dehalogenative, dehydrogenative and defluorinative coupling, and provides us a rational way for synthesizing cross-linked functional materials.


 Please wait while we load your content...
Please wait while we load your content...