Dynamic parallel kinetic resolution of α-ferrocenyl cation initiated by chiral Brønsted acid catalyst†
Abstract
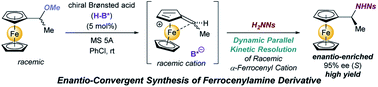
The dynamic parallel kinetic resolution (DPKR) of an α-ferrocenyl cation intermediate under the influence of a chiral conjugate base of a chiral phosphoric acid catalyst has been demonstrated in an SN1 type substitution reaction of a racemic ferrocenyl derivative with a nitrogen nucleophile. The present method provides efficient access to a ferrocenylethylamine derivative in a highly enantioselective manner, which is potentially useful as a key precursor of chiral ligands for metal catalysis. The mechanism of the present intriguing resolution system was elucidated by control experiments using the enantio-pure precursor of relevant α-ferrocenyl cation intermediates and the hydroamination of vinylferrocene. Further theoretical studies enabled the elucidation of the origin of the stereochemical outcome as well as the efficient DPKR. The present DPKR, which opens a new frontier for kinetic resolution, involves the racemization process through the formation of vinylferrocene and the chemo-divergent parallel kinetic resolution of the enantiomeric α-ferrocenyl cations generated by the protonation/deprotonation sequence of vinylferrocene.


 Please wait while we load your content...
Please wait while we load your content...