The dioxasilepanyl group as a versatile organometallic unit: studies on stability, reactivity, and utility†
Abstract
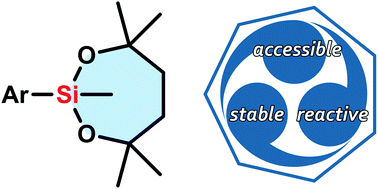
Organic synthesis is performed based on precise choices of functional groups and reactions employed. In a multistep synthesis, an ideal functional group should be compatible with various reaction conditions and unaltered until it is subjected to a selective conversion. The current study was set out to search for a silicon functionality that meets these criteria. Here we have established a new silicon-based synthetic methodology centred on a bulky 7-membered dialkoxysilyl group (2,4,4,7,7-pentamethyl-1,3,2-dioxasilepan-2-yl) that uniquely has both stability and on-demand reactivity. The exceptional stability of this functional group was corroborated by both experimental and computational studies which demonstrated that key factors for its stability were a 7-membered structure and steric hindrance. In turn, the dioxasilepanyl group was found to become reactive and to be easily transformed in the presence of appropriate activators. Combined with the development of easy and robust methods to introduce the dioxasilepanyl group onto aryl rings, these findings have allowed a shorter and more efficient synthesis of a bioactive molecule, thus demonstrating the potential utility of the easily accessible dioxasilepanyl group in organic synthesis.


 Please wait while we load your content...
Please wait while we load your content...