Probing conformational hotspots for the recognition and intervention of protein complexes by lysine reactivity profiling†
Abstract
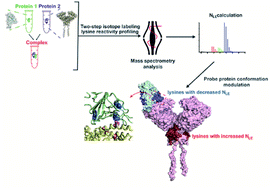
Probing the conformational and functional hotspot sites within aqueous native protein complexes is still a challenging task. Herein, a mass spectrometry (MS)-based two-step isotope labeling-lysine reactivity profiling (TILLRP) strategy is developed to quantify the reactivities of lysine residues and probe the molecular details of protein–protein interactions as well as evaluate the conformational interventions by small-molecule active compounds. The hotspot lysine sites that are crucial to the SARS-CoV-2 S1–ACE2 combination could be successfully probed, such as S1 Lys417 and Lys444. Significant alteration of the reactivities of lysine residues at the interaction interface of S1-RBD Lys386–Lys462 was observed during the formation of complexes, which might be utilized as indicators for investigating the S1-ACE2 dynamic recognition and intervention at the molecular level in high throughput.
- This article is part of the themed collection: Coronavirus articles - free to access collection


 Please wait while we load your content...
Please wait while we load your content...