Conformational editing of intrinsically disordered protein by α-methylation†
Abstract
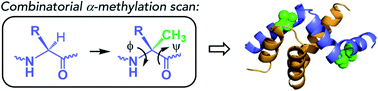
Intrinsically disordered proteins (IDPs) constitute a large portion of “Dark Proteome” – difficult to characterize or yet to be discovered protein structures. Here we used conformationally constrained α-methylated amino acids to bias the conformational ensemble in the free unstructured activation domain of transcriptional coactivator ACTR. Different sites and patterns of substitutions were enabled by chemical protein synthesis and led to distinct populations of α-helices. A specific substitution pattern resulted in a substantially higher binding affinity to nuclear coactivator binding domain (NCBD) of CREB-binding protein, a natural binding partner of ACTR. The first X-ray structure of the modified ACTR domain - NCBD complex visualized a unique conformation of ACTR and confirmed that the key α-methylated amino acids are localized within α-helices in the bound state. This study demonstrates a strategy for characterization of individual conformational states of IDPs.


 Please wait while we load your content...
Please wait while we load your content...