Zika virus lateral flow assays using reverse transcription-loop-mediated isothermal amplification†
Abstract
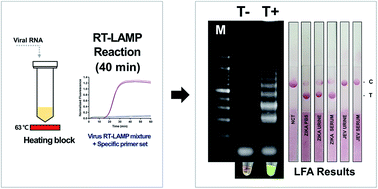
Owing to the global spread of the Zika virus (ZIKV) infection, field-ready diagnostics are urgently warranted. In this study, we sought to detect ZIKV using reverse transcription loop-mediated isothermal amplification (RT-LAMP). Briefly, we performed and optimized ZIKV RT-LAMP for the analysis of biological samples (PBS, urine, and plasma). Based on our findings, this method could detect ZIKV RNA in 40 min at 63 °C without any off-target amplification. After performing specificity tests using BtsI restriction enzyme digestion, the feasibility of ZIKV RT-LAMP was determined via end-point detection with different sample matrices. Thereafter, a lateral flow assay (LFA) was conducted to directly detect the ZIKV RT-LAMP products. Based on the LFA reaction, hybridization occurred between the AuNPs:polyadenylated (polyA10)-ZIKV probe and the LAMP amplicons. Subsequently, we optimized the assay parameters, including the concentration of AuNPs and migration matrices (glass fiber and nitrocellulose membrane). By employing a specific AuNP:polyA10-ZIKV LAMP probe, we could demonstrate the purpose and utility of primary and secondary antibodies. Owing to LFA, the resultant ZIKV RT-LAMP products were rapidly and simply assayed in less than 5 min. Further, no preparation step was required to achieve LAMP-probe hybridization, highlighting the utility of this method for field-ready ZIKV diagnosis. Collectively, our findings suggest that ZIKV RT-LAMP combined with LFA could serve as a rapid, accurate, and independent point-of-care detection method for preventing ZIKV outbreaks.


 Please wait while we load your content...
Please wait while we load your content...