Highly sensitive fluorescence detection of tobacco mosaic virus RNA based on disodium 4,4′-diazidostilbene-2,2′-disulfonate tetrahydrate in situ reaction
Abstract
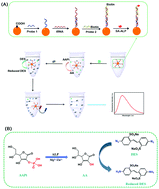
A highly sensitive fluorescence detection method for tobacco mosaic virus (TMV) RNA based on in situ reaction of disodium 4,4′-diazidostilbene-2,2′-disulfonate tetrahydrate (DES) was developed for the first time. Specifically, cDNA-1 (probe 1) was fixed on carboxylated Fe3O4 magnetic beads (MBs), and hybridized with the TMV RNA (tRNA) in the next step. Then, cDNA-2 (probe 2) that was hybridized with tRNA was linked to alkaline phosphatase-conjugated streptavidin (SA-ALP) via the binding of biotin and SA. Next, L-ascorbic acid 2-phosphate sesquimagnesium salt hydrate (AAPi) was dephosphorylated and hydrolyzed by alkaline phosphatase (ALP), producing ascorbic acid (AA). The subsequently added DES was reduced by AA, producing reduced DES, which successfully resulted in ‘turn-on’ of the fluorescent signal. Under optimal conditions, a good linear relationship between the fluorescence intensity and the logarithm of the tRNA concentration was obtained in the range from 1 pM to 10 nM, with a detection limit of 0.101 pM. Meanwhile, the method exhibited excellent selectivity and reproducibility. These results showed that the fluorescence detection strategy can be effectively employed for the qualitative and quantitative analysis of tRNA and has a potential application in the actual detection of tRNA.


 Please wait while we load your content...
Please wait while we load your content...