2D g-C3N4 as a bifunctional photocatalyst for co-catalyst and sacrificial agent-free photocatalytic N2 fixation and dye photodegradation†
Abstract
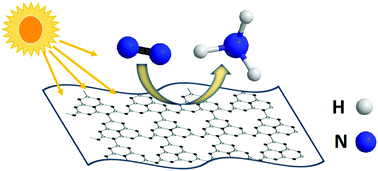
Photocatalytic N2 fixation is an ecofriendly technology to produce ammonia. In this work, photocatalytic N2 fixation to generate ammonia in the absence of any co-catalysts or sacrificial agents (SF) is achieved on 2D g-C3N4 prepared via thermal exfoliation, for the first time. The formation of 2D g-C3N4 is confirmed using several analytical techniques including XRD, SEM, TEM, FT-IR, and XPS analyses. 2D g-C3N4 exhibits a blue shift in the bandgap compared to the bulk g-C3N4 attributed to the quantum confinement effect. In the absence of SF, 2D g-C3N4 shows an NH3 production rate of 3.9 mg L−1 h−1 under sunlight irradiation, while no NH3 is generated on bulk g-C3N4. In the presence of ethanol as a sacrificial agent, 2D g-C3N4 also exhibits a higher NH3 production rate (12.76 mg L−1 h−1) than bulk g-C3N4 (7.58 mg L−1 h−1). Moreover, 2D g-C3N4 shows superior photodegradation activity compared to bulk g-C3N4 toward the decomposition of methylene blue (MB) and malachite green (MG) under sunlight irradiation. The enhanced photocatalytic activity of 2D g-C3N4 could be attributed to its more exposed active sites, high charge separation, and low electron–hole recombination rate.


 Please wait while we load your content...
Please wait while we load your content...