Catalyst-free synthesis of phenanthridines via electrochemical coupling of 2-isocyanobiphenyls and amines†
Abstract
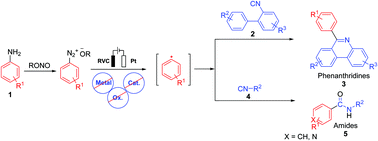
Catalyst free synthesis of 6-aryl phenanthridines and amides through an electrochemical reaction is reported in this study. The coupling reaction proceeds by the cathodic reduction of in situ formed diazonium ions, which are formed from anilines and an alkyl nitrite. The generated aryl radical diazonium ions coupled from isocyanides furnished the desired products in good yields. This cascade reaction was conducted in an undivided cell equipped with an RVC as the anode and Pt as the cathode using nBu4NBF4 as the electrolyte at room temperature. A series of detailed mechanistic studies have also been performed, including a radical clock experiment and cyclic voltammetry analysis.


 Please wait while we load your content...
Please wait while we load your content...