Nickel oxide regulating surface oxygen to promote formaldehyde oxidation on manganese oxide catalysts†
Abstract
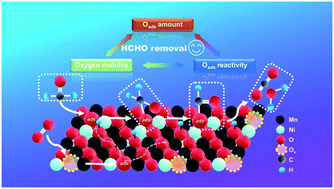
Catalytic oxidation is the most effective method to eliminate in-door formaldehyde, and Mn-based catalysts with low cost and high activity have drawn great attention for this reaction. Herein, p-type semiconductor NiO doped MnOx catalysts were prepared by an environmentally friendly oxalate co-precipitation method. The doped Ni species entered the lattice of MnOx to form amorphous Ni–Mn composite oxide/NiO and increased the ratio of surface Mn4+ and total amount of surface active oxygen simultaneously with the increase of NiO content in the form of a volcano curve, which directly correlated to the abilities for adsorption and oxidation of formaldehyde. Among them, 0.2NiO–MnOx (Ni/(Ni + Mn) = 0.2) showed the highest activity, 300 ppm of formaldehyde can be completely eliminated at 98 °C in a 2.5 vol% H2O-containing atmosphere, and the corresponding specific reaction rate was about 2.9 times higher than that of pure MnOx. Meanwhile, the enhanced migration of oxygen species over NiO–MnOx catalysts also promoted the replenishment of surface active oxygen that was consumed in the reaction process, which kept the activity of 0.2NiO–MnOx stable during the continuous reaction cycles and 26 h long-term stability test. Our study showed that utilizing NiO to regulate the reactivity and amount of surface oxygen species was an efficient way to improve the intrinsic catalytic performance of MnOx.


 Please wait while we load your content...
Please wait while we load your content...