Probing into the double identity of Cu in bimetallic CuAg-TNTA hot-electron devices for photo-hydrogen conversion†
Abstract
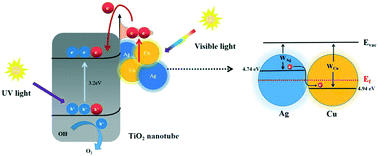
Bimetallic nanostructures have been endowed with the function of enhancing photon absorption and charge separation for photo-hydrogen conversion. Bimetallic CuAg/TiO2 nanotube arrays (TNTAs) were synthesized with the different ratios of precursors Cu2+ and Ag+ (1 : 3, 1 : 1, and 3 : 1) and their local electric field distribution was characterized by means of COMSOL software. The results demonstrate that the increase of the amount of Cu particles has an active effect on the inhibition of the hot spot effect between Ag particles. In addition, the current flow between Ag and Cu verified the “double take” of Cu particles. They receive hot electrons transferred from Ag, and also provide injected hot electrons for TiO2. Based on X-ray photoelectron spectroscopy, the binding energy shifts in a CuAg bimetal with different metal ratios, indicating the electron transfer behavior from Ag to Cu. When an optimal proportion of the CuAg bimetal is 3 : 1, the photocurrent density and average hydrogen evolution rate of CuAg/TNTAs were 1.78 and 1.69 times higher, respectively, than those of pure TNTAs, implying far more efficient migration of hot electrons.


 Please wait while we load your content...
Please wait while we load your content...