Recognition of hydrophilic molecules in deep cavitand hosts with water-mediated hydrogen bonds†
Abstract
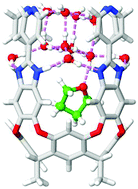
We describe new container host molecules – deep cavitands with benzimidazole walls and ionic feet – to recognize highly hydrophilic guest molecules in water. The aromatic surfaces of the cavity recognize hydrophobic portions of the guest while bound water molecules mediate hydrogen bonding in the complex. Spectroscopic (NMR) evidence indicates slow in/out exchange on the chemical shift timescale and thermodynamic (ITC) methods show large association constants (Ka up to 6 × 104 M−1) for complexation of small, water-soluble molecules such as THF and dioxane. Quantum chemical calculations are employed to optimize the host–guest geometries and elucidate the hydrogen bonding patterns responsible for the binding.
- This article is part of the themed collection: Host-Guest Chemistry


 Please wait while we load your content...
Please wait while we load your content...