Controlling fluorescence resonance energy transfer of donor–acceptor dyes by Diels–Alder dynamic covalent bonds†
Abstract
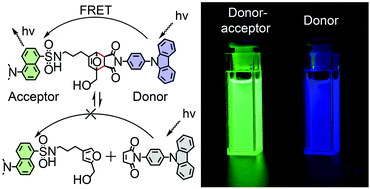
Two new dyes consisting of an aromatic amine donor and dansyl acceptor were synthesized, where the intramolecular donor and acceptor are connected by Diels–Alder dynamic covalent bonds. These new dyes display a switchable fluorescence resonance energy transfer through reversible formation and cleavage of Diels–Alder bonds. Single crystal X-ray diffraction revealed that Diels–Alder bonds are longer and weaker than normal single bonds. Dynamic covalent properties enable the mutual conversion of the two dyes by maleimide exchanges, where a new higher energy transfer efficiency system can be constructed.


 Please wait while we load your content...
Please wait while we load your content...