Effect of new carbonyl cyanide aromatic hydrazones on biofilm inhibition against methicillin resistant Staphylococcus aureus†
Abstract
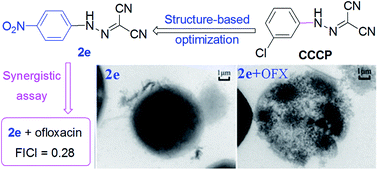
Carbonyl cyanide m-chlorophenylhydrazone (CCCP), as a protonophore, in combination with antibiotics exhibited potentiating antibacterial activity. To improve CCCP's potency and toxicity, a series of aromatic hydrazones were synthesized and their antimicrobial activity was evaluated; amongst them, compounds 2e and 2j with a strong para-electron-withdrawing substituent (–NO2 and –CF3) at the phenyl ring had the lowest MICs against both S. aureus and methicillin resistant Staphylococcus aureus (1.56 and 1.56 μM, respectively). Some compounds in combination with antibiotics exhibited potentiate Gram-positive antibacterial activity; compound 2e was found to display unaided or synergistic efficacy against MRSA. In particular, when compound 2e is combined with ofloxacin, it has a good synergistic effect against MRSA. Moreover, electron microscopy revealed that compound 2e inhibits biofilm formation and effectively eradicates preformed biofilm. MTT assay showed that compound 2e displays as low toxicity as CCCP. Overall, our data showed that the aromatic hydrazone is a promising scaffold for anti-staphylococcal drug development.


 Please wait while we load your content...
Please wait while we load your content...