Quantum chemical (QM:MM) investigation of the mechanism of enzymatic reaction of tryptamine and N,N-dimethyltryptamine with monoamine oxidase A†
Abstract
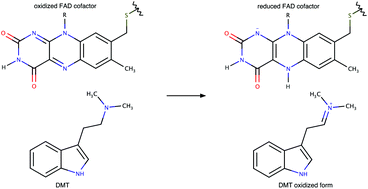
The endogenous psychedelic (mind-altering) N,N-dimethyltryptamine (DMT) molecule has an important role in tissue protection, regeneration, and immunity via sigma-1 receptor activation as its natural ligand. The immunologic properties of DMT suggest this biogenic compound should be investigated thoroughly in other aspects as well. In our in silico project, we examined the metabolism of DMT and its primary analogue, the tryptamine (T), by the monoamine oxidase (MAO) flavoenzyme. MAO has two isoforms, MAO-A and MAO-B. MAOs perform the oxidation of various monoamines by their flavin adenine dinucleotide (FAD) cofactor. Two-layer QM:MM calculations at the ONIOM(M06-2X/6-31++G(d,p):UFF=QEq) level were performed including the whole enzyme to explore the potential energy surface (PES) of the reactions. Our findings reinforced that a hybrid mechanism, a mixture of pure H+ and H− transfer pathways, describes precisely the rate-determining step of amine oxidation as suggested by earlier works. Additionally, our results show that the oxidation of tertiary amine DMT requires a lower activation barrier than the primary amine T. This may reflect a general rule, thus we recommend further investigations. Furthermore, we demonstrated that at pH 7.4 the protonated form of these substrates enter the enzyme. As the deprotonation of substrates is crucial, we presumed protonated cofactor, FADH+, may form. Surprisingly, the activation barriers are much lower compared to FAD with both substrates. Therefore, we suggest further investigations in this direction.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...