A high-capacity iron silicide–air primary battery in an acidic saline electrolyte†
Abstract
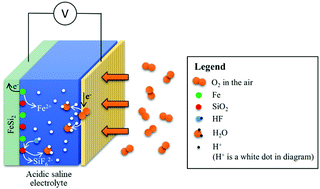
Iron silicide is widely used in the steel and metallurgical industry, and it shows anti-corrosion performance in acidic solution. In this paper, we report an FeSi2–air battery in an acidic saline electrolyte for the first time. The usage of the FeSi2 anode in acidic electrolyte can overcome the limitations of the Fe anode in alkaline electrolyte, such as the formation of a passive layer on the surface of the anode, low solubility of Fe(OH)2, etc. In this paper, we are the first to assemble an FeSi2–air battery in acidic saline electrolyte, which showed a high specific capacity of 1.90 A h g−1 in 0.5 M H2SO4 saline electrolyte in the presence of F− ions, which was close to approximately 80% of the theoretical capacity. Control experiments were carried out to propose possible mechanisms of the discharge of the FeSi2 anode in acidic saline electrolyte. In the presence of F− ion in acidic saline electrolyte, a sustainable discharge process can last for a long period until the active material in the anode is used up. Furthermore, it shows an excellent utilization of both gravimetric capacity and volumetric capacity compared with other metal, metalloid and metal/metalloid–air batteries. Considering the less pollution of the environment, the FeSi2–air battery will be a competitive battery with high specific capacity and utilization for further applications.


 Please wait while we load your content...
Please wait while we load your content...