Size dependent oxygen reduction and methanol oxidation reactions: catalytic activities of PtCu octahedral nanocrystals†
Abstract
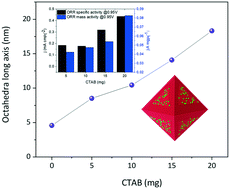
The synthetic control through colloidal synthesis led to a remarkable increase in platinum mass activity in octahedral nanocrystals with a Pt-rich surface. In this manuscript, we demonstrate that the ratio of surfactant can tune the size of Pt surface enriched PtCu nano-octahedra from 8 to 18 nm with homogeneous size and shape on the carbon support. For the nano-octahedra, the Pt-rich surface has been determined by high-angle annular dark field scanning transmission electron microscopy and energy-dispersive X-ray spectroscopy. The Pt-rich surface exhibits an increasing compressive strain with increasing surface of the {111} facets. With increasing surface, the PtCu nano-octahedra display higher oxygen reduction reaction (ORR) activity, which however leads to higher onset over-potentials in the methanol oxidation reaction (MOR) and CO-stripping. This observed trend for a series of size-selected nano-octahedra demonstrates the benefits of controlling the strained {111} Pt surface for the ORR and MOR activity.


 Please wait while we load your content...
Please wait while we load your content...