Pt (111) quantum dot decorated flower-like αFe2O3 (104) thin film nanosheets as a highly efficient bifunctional electrocatalyst for overall water splitting†
Abstract
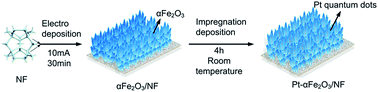
Water splitting is a promising approach for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER). The search for highly active and cost-effective bifunctional catalysts to reduce the consumption of noble metals is necessary for efficient electrochemical water splitting. Using noble metal quantum dots has drawn increasing attention. Herein, we developed flower-like Pt–αFe2O3 thin film nanosheets on Ni foam (NF) as a highly efficient bifunctional electrocatalyst for overall water splitting in alkaline electrolyte. X-ray diffraction (XRD), field-emission scanning electron microscopy (FESEM), transmission electron microscopy (TEM), atomic force microscopy (AFM), energy-dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectroscopy (XPS) techniques were used to characterize the electrocatalyst. The unique design of Pt (111) quantum dot decorated flower-like αFe2O3 (104) thin film nanosheets exhibited a quantum size effect and possessed many active sites. Pt–αFe2O3/NF electrodes which were loaded with a small amount of Pt quantum dots exhibited low overpotentials for the OER (304 mV@50 mA cm−2) and HER (90 mV@10 mA cm−2), and a low voltage of 1.51 V for overall water splitting in 1 M KOH. Moreover, excellent stability was observed. Therefore, the flower-like nanosheet Pt–αFe2O3/NF electrode is a promising bifunctional electrocatalyst with the quantum size effect for overall water splitting in alkaline solution.


 Please wait while we load your content...
Please wait while we load your content...