A molecular approach on the ability of functionalized single walled carbon nanotube for cathinone sensing
Abstract
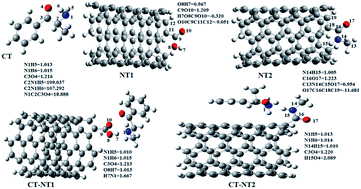
In this article, single walled carbon nanotube functionalized with COOH (NT1) and CONHCH3 (NT2) groups were used for detection of the cathinone (CT) molecule in the gas phase and the liquid phase from the theoretical point of view. Density functional theory (DFT) calculations indicate that the NT2 nanostructure is more sensitive to the cathinone molecule than to the NT1 nanostructure. Compared to the gas phase, in the liquid phase water increases the sensitivity of the nanostructures toward the cathinone molecules. DFT results show that the polarity of the solvent increases the stability of the complexes. Donor–acceptor orbital interactions reveal that the cathinone molecule has a more effective orbital interaction with the NT2 nanostructure, especially in a water solvent. Also, molecular dynamic (MD) simulations confirm that the interactions between the cathinone molecule and the nanostructures increase in the water solvent. Therefore, NT nanostructures are more sensitive toward the CT molecule in a water solvent.


 Please wait while we load your content...
Please wait while we load your content...