Theoretical studies on the purine radical induced purine–purine type intrastrand cross-links†
Abstract
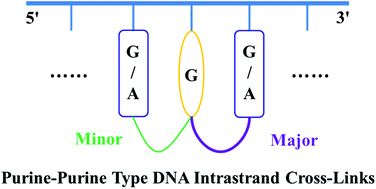
At the density functional theory (DFT) level, addition reactions between the guanine-8-yl radical and its 3′/5′ neighboring purine deoxynucleosides forming the purine–purine type intrastrand cross-links were studied. It is found that addition of the guanine-8-yl radical to the C8 site of its 5′ neighboring deoxyguanosine or deoxyadenosine is a two-step reaction consisting of a structurally relatively unfavourable conformational transformation step, while the corresponding 3′ C8 addition is straightforward and kinetically more efficient. The 3′ C8 preference of the guanine-8-yl radical additions indicates the existence of an obvious sequence effect, which is completely opposite to that observed in the formation of pyrimidine radicals induced DNA intrastrand cross-links. The detrimental effects from steric hindrance and stabilizing weak interactions make these addition reactions markedly suppressed in double stranded DNA. This work broadens our knowledge about the possible types of DNA intrastrand cross-links.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...