The QM/MM-QTAIM approach reveals the nature of the different reactivity of cephalosporins in the active site of L1 metallo-β-lactamase†
Abstract
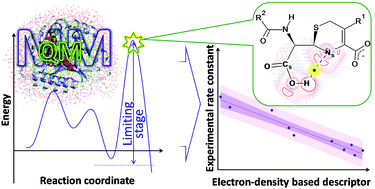
We developed relationships between the calculated microscopic parameters in the active site of L1 metallo-β-lactamase and the experimentally observed catalytic rate constants kcat of hydrolysis of cephalosporin compounds. The atomic interactions are quantified using various QTAIM bond descriptors in the active sites of 10 protein–antibiotic complexes obtained at the QM/MM level in the transition states of the limiting stage. We identify key atomic interactions that are mainly responsible for the macroscopic properties and reveal that the Ns⋯H hydrogen bond determines the catalytic rate constants in cephalosporin–L1 metallo-β-lactamase systems. The best descriptor for this interaction is the contribution of the Ns atom to the electron density at the hydrogen bond critical point, which was established using the source function for the electron density. The estimated rate constant for ceftazidime, which was not included in the training set, is 15.7 ± 3.9 s−1,which reproduces the experimental kcat value of 15.4 s−1. Also, we propose a fluorinated derivative of cefoxitin that demonstrates the lowest rate constant among the known cephalosporin compounds. We suppose that our concept is transferable for other enzymatic reactions.


 Please wait while we load your content...
Please wait while we load your content...