Tunable physicochemical and free volume characteristics of novel terpolymer–poly(vinyl alcohol)-grafted membranes for direct methanol fuel cells
Abstract
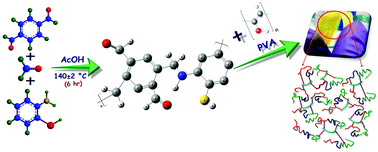
Here, we describe a facile and scalable approach to fabricate novel ABC-type terpolymer-based proton conducting membranes (PCMs) and elucidate the benefits of utilizing terpolymer composite membrane polymer materials as electrolytes for direct methanol fuel cells (DMFC). A novel terpolymer has been synthesized from terephthaldehyde, 2-aminothiophenol and formaldehyde (TAF) by condensation polymerization under optimized conditions. The chemical structure, thermal stability, size and morphology of the synthesized TAF terpolymer were studied by various techniques, such as scanning electron microscopy (SEM), energy dispersive X-ray spectroscopy (EDX), Proton nuclear magnetic resonance spectroscopy (1H NMR), Fourier transform infrared spectroscopy (FT-IR), thermogravimetric analysis and X-ray diffraction (XRD) analysis. Solution casting using varied amounts of this novel cross-linkable terpolymer allowed the tuning of multiple interfaces to produce composite membranes with improved properties. These membranes and their preliminary electrochemical properties (ionic exchange capacity, proton conductivity, and swelling rate) were studied and optimized based on proton conductance. In addition, positron annihilation spectroscopic techniques were used to characterize the sizes and concentrations of free volume defects in the samples, which can significantly alter the physical and thermodynamic properties of these complex polymer materials.


 Please wait while we load your content...
Please wait while we load your content...