Chemo-enzymatic pathways toward pinene-based renewable materials†
Abstract
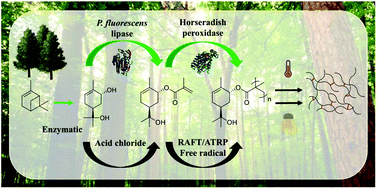
Sobrerol methacrylate (SobMA) was synthesized and subsequently polymerized using different chemical and enzymatic routes. Sobrerol was enzymatically converted from α-pinene in a small model scale by a Cytochrome P450 mutant from Bacillus megaterium. Conversion of sobrerol into SobMA was performed using both classical ester synthesis, i.e., acid chloride-reactions in organic solvents, and a more green approach, the benign lipase catalysis. Sobrerol was successfully esterified, leaving the tertiary alcohol and “ene” to be used for further chemistry. SobMA was polymerized into PSobMA using different radical polymerization techniques, including free radical (FR), controlled procedures (Reversible Addition Fragmentation chain-Transfer polymerization, (RAFT) and Atom Transfer Radical Polymerization (ATRP)) as well as by enzyme catalysis (horseradish peroxidase-mediated free radical polymerization). The resulting polymers showed high glass-transition temperatures (Tg) around 150 °C, and a thermal degradation onset above 200 °C. It was demonstrated that the Tg could be tailored by copolymerizing SobMa with appropriate methacrylate monomers and that the Flory–Fox equation could be used to predict the Tg. The versatility of PSobMA was further demonstrated by forming crosslinked thin films, either using the ‘ene’-functionality for photochemically initiated ‘thiol-ene’-chemistry, or reacting the tertiary hydroxyl-group with hexamethoxymethylmelamine, as readily used for thermally curing coatings systems.


 Please wait while we load your content...
Please wait while we load your content...