Selective hydrodeoxygenation of lignin oil to valuable phenolics over Au/Nb2O5 in water†
Abstract
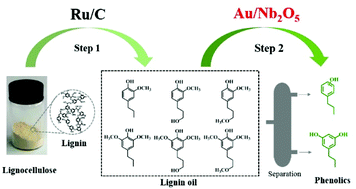
Transformation of lignin into high value-added phenolics is of great significance but extremely challenging. Herein, we report a single-step process for the preparation of phenolics from both 2-methoxy-4-propylphenol (one of the major products derived from lignin) and lignin oil (lignin-derived monomers) over Au-based catalysts. In this process, the desired Ar–OH substituents can be maintained and phenolics can be obtained with high yields. The performance of the Au/Nb2O5 catalyst is compared with those of Au/TiO2, Au/Al2O3 and Au/ZrO2 and it is shown that Au/Nb2O5 has high activity, excellent stability and remarkable selectivity to phenolics, with the yields of phenolics reaching 84.2% for 2-methoxy-4-propylphenol and 66.7 wt% for lignin oil. The detailed studies reveal that the hydrodeoxygenation of 2-methoxy-4-propylphenol undergoes two-steps, i.e., hydrolysis to propylcatechol catalyzed by the acid sites of supports, and the hydrogenolysis of propylcatechol to propylphenol realized by the synergistic effect between Au and the supports. It is found that the high hydrodeoxygenation activity of Au/Nb2O5 depends on both the electron-rich Au nanoparticles and the Nb2O5 support with the efficient cleavage of the Caromatic–O linkage. This study provides a new catalytic system towards highly efficient and selective conversion of lignin oils to valuable phenolics.


 Please wait while we load your content...
Please wait while we load your content...